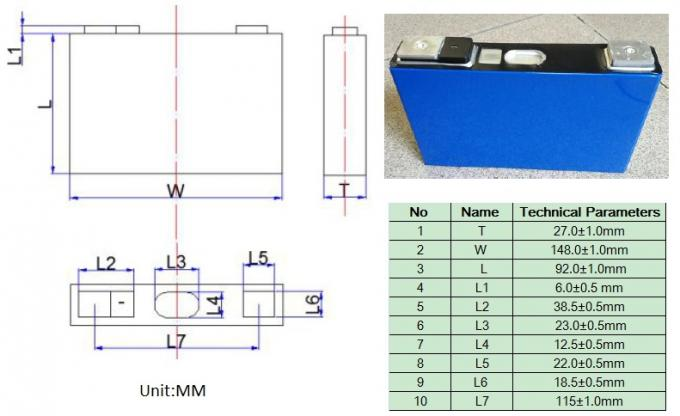
NMC batteries technology and applications
In 2008, the NMC (Nickel Manganese Cobalt Oxide) battery was developed. LiaC6 serves as the anode, LibNi1–x–yCoxMnyO2 serves as the cathode, and Li+ serves as the carrier. It is frequently utilized in a variety of applications, including energy systems and electric automobiles. The best of manganese and nickel are combined in NMC. Nickel has a low stability but a high specific energy; Manganese has the ability to form a spinel structure with low specific energy and low internal resistance. The strengths of both parts are increased when the metals are joined. Power tools, energy storage systems (EES), and electrical powertrains like automobiles make extensive use of NMC batteries. Typically, the cathode composition is 1-1-1, or one third nickel, one third manganese, and one third cobalt. Due to the lower cobalt content, this lowers the cost of the raw materials.
NMC is the battery of choice for power tools, e-bikes and other electric powertrains. The cathode combination is typically one-third nickel, one-third manganese and one-third cobalt, also known as 1-1-1. Cobalt is expensive and in limited supply.
What is NMC battery?
NMC stands for Lithium Nickel Manganese Cobalt Oxide, which is a type of rechargeable battery commonly used in electric vehicles and other high-performance applications. NMC batteries offer high energy density, good power performance, and long cycle life, making them a popular choice for many applications.
What is difference between NMC and LFP battery?
The main difference between NMC and LFP batteries lies in their chemical composition and performance characteristics. NMC batteries use a combination of nickel, manganese, and cobalt in their cathode material, while LFP batteries use lithium iron phosphate.
In terms of performance, NMC batteries typically have a higher energy density and offer better power performance, which makes them a popular choice for electric vehicles and other high-performance applications. LFP batteries, on the other hand, have a longer cycle life and are more resistant to thermal runaway, making them a safer option for stationary energy storage and other applications where safety is a primary concern.

Are NMC batteries good?
For applications where space is the primary concern, NMC batteries are beneficial because of their high energy density. Therefore, NMC batteries are the better choice for applications such as electronics, mobile phones, laptops, etc.
Which battery is better lithium ion or NMC?
Even at higher temperatures, the Lithium Iron Phosphate chemistry is more stable than Nickel Manganese Cobalt. At the same time, LFP batteries are less likely to go under thermal runaway than NMC batteries due to their ability to handle larger power draws. Hence, LFP batteries are less vulnerable to catching fire.
Are Tesla batteries NMC?
Tesla announced in October 2021 that it was switching to LFP batteries for its standard-range models (Model 3 and Model Y), while retaining the NMC cells for longer-range models.
What battery is better LFP or NMC Tesla?
LFP batteries don’t contain nickel, cobalt and magnesium, resulting in cheaper manufacturing costs. It’s also more robust and less susceptible to thermal runaway compared to NMC. A key advantage of LFP is its longer life cycle, resulting in less degradation concerns.
Is Tata Nexon battery NMC or LFP?
The Tata Nexon EV uses Lithium ferrous phosphate (LFP) cells, as LFP has more ability for thermal stress than nickel-manganese-cobalt (NMC) cells. The Nexon EV battery is made in India.

What is the life cycle of NMC battery?
As a default, both NMC and LFP chemistries’ useful life can range between 3,000 to 5,000 cycles. However, with opportunity charging, that can be increased significantly, anywhere up to 7,000 cycle count.
Advantages and disadvantages of NMC batteries:
The NMC batteries have the following main advantages:
NMC cells have a longer life cycle, are cheaper, and have a higher energy density. By weight and volume, NMC cathodes contain the most energy.
The following are the primary drawbacks of NMC batteries:
The disadvantage of adding silicon to graphite is that it causes the anode to expand and contract as the cell charges and discharges, resulting in mechanical instability.
less voltage than cobalt systems
What is the disadvantage of NMC battery?
The main disadvantages of the NMC batteries are:
- Adding silicon to graphite has the downside of making the anode grow and shrink through charging and discharging, leading to mechanical instability of the cell.
- Slightly lower voltage than cobalt systems.
Conclusion;
NMC batteries are a type of rechargeable battery with high energy density and long cycle life, used in electric vehicles and energy storage systems. However, they have a tendency to experience thermal runaway and are relatively expensive.




